The Shapes of Molecules
To make the shapes of the molecules named above as realistic as possible, the angles between the H's should be right. Methane's H's are at the 4 points of a tetrahedron, and ammonia and water are very similar (but with some vertices not having H's).
You can print out a cardboard guide showing where to stick the toothpicks into the styrofoam spheres by folding a cardboard tetrahedron of just the right size (2 x 61/2 times the diameter of the sphere) around the sphere,...then skewer it through the centers of the bottom face and 1, 2, or all 3 of the other faces, resulting in something like the photos on the right. You may need to print the guide larger or smaller, depending on the size of your styrofoam sphere. (Note the slit for folding the face away from the sphere without having to remove the toothpick.)
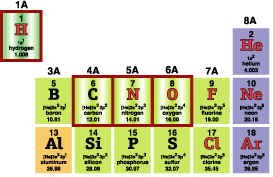
For Oxygen, only 2 toothpicks would be used, for Nitrogen, 3; if double bonds are planned, pull out the toothpicks involved and replace with pipecleaner segments. |


|
![]()